Astatos definition3/25/2023  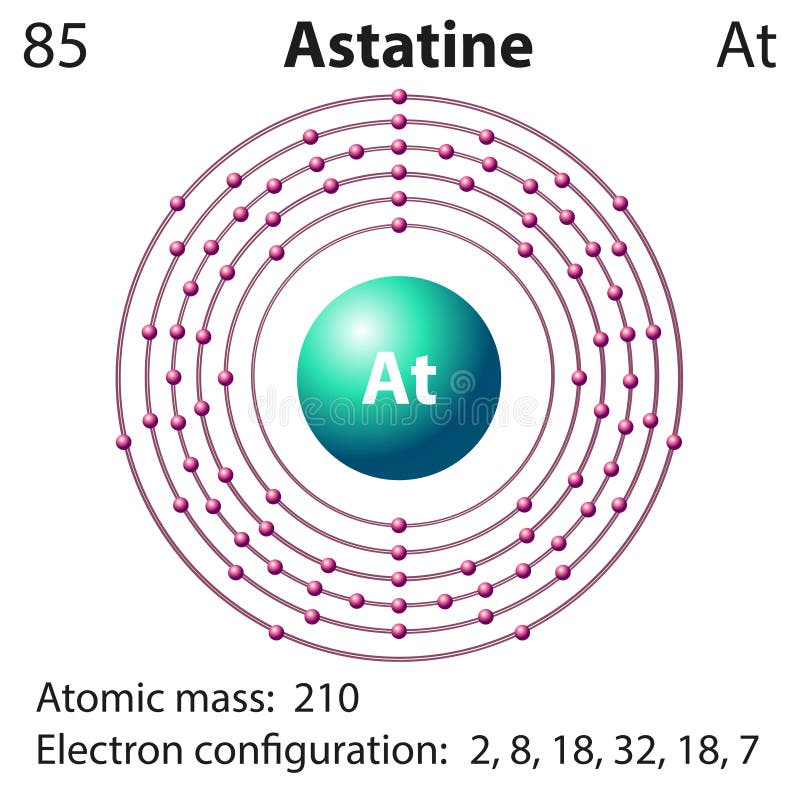
Note: This article, originally published in 1998, was updated in 2006 for the eBook edition. "It's Elemental: Astatine." Jefferson Lab. The CRC Handbook of Chemistry and Physics, 84th edition. Leave your influence in your wake as you bring together a broken land in a card-battling adventure filled with gods, heroes, and the instability of yourself and those around you. 
Because of its similarities to iodine, which accumulates in the thyroid, it is believed that the element could be utilized to treat certain thyroid diseases. A unique blend of engaging narrative and tabletop-inspired card action about the influence you have on others. This means the Studio Klondike Expansion Pack (IV) will be included with every backers copy of Astatos Adding six new hero cards based on characters from. Researchers are investigating astatine as a means of treating various cancers (e.g., lethal brain tumors) and diseases. Astatos is a card-battling adventure filled with gods, heroes, and the instability of yourself and those around you. Additional compounds (HAt and CH 3At) have also been detected.īecause of its scarcity and short half-life, there were no commercial uses for astatine as of 2003. Astatine is known to form interhalogen compounds with bromine (AtBr), chlorine (AtCl), and iodine (AtI). Astatine, At, Greek astatos, meaning unstable. Other isotopes have mass numbers ranging from 193 to 223 and half-lives ranging from 125 nanoseconds ( 213At) to 7.2 hours ( 211At). Antimony, Sb, Greek anti monos, meaning not alone Latin stibium, the name of antimony sulfide in. The most stable isotope of astatine is 210At, which has a half-life of 8.1 hours. Astatine can also be produced in a nuclear reactor by the method used by its discoverers, according to the following reaction: It is produced naturally when the elements uranium and thorium decay. All of astatine's isotopes are short-lived the most stable is astatine-210, with a half-life of 8.1 hours. It is the rarest naturally occurring element in the Earth's crust, occurring only as the decay product of various heavier elements. The origin of the name "astatine" is the Greek word astatos, which means "unstable."Īstatine is found in only vanishingly small amounts in nature -it is believed that only 30 grams (1 ounce) of the element are present in Earth's crust at any one time. Astatine is a chemical element with the symbol At and atomic number 85. MacKenzie, and Emilio Segr è of the University of California produced the element in 1940 by bombarding an isotope of bismuth ( 209Bi) with alpha particles. The two atoms form a covalent bond by sharing two electrons between them.Astatine is a radioactive halogen (the heaviest of the halogen elements) and is a solid at room temperature. FluorineĪll halogens exist as diatomic molecules or in other words as two- atom molecules. Scientists tried finding the element that followed iodine in the halogen group of the. see how the trend of cash shops have been the past few years, theres a good reason to be concerned. 
Chlorine molecules have the formula C l 2, bromine Br 2 and iodine I 2. Astatine comes from the Greek word astatos, meaning unstable. oh i know the cash shop is optional, it just worries me. This means they exist as molecules, each with a pair of atoms. The halogens are so reactive that they cannot exist free in nature. In general the halogens comprise the most reactive group of non-metals. The term 'halogen' means ' salt former', which is why Group 7 elements are called halogens. Group 7 elements form salts when they react with metals. The name Astatine is from the Greek word ' astatos' meaning unstable. Its most stable isotope 240 85 At has a half-life of 8.3 hours. It is a very unstable, radioactive element first synthesised in 1940 by chemist in the USA. Fluorine, chlorine, bromine, iodine, and astatine, all belong to Group 7.Īstatine (At), the last element in the Group does not occur naturally. All Group 7 elements have 7 electrons in their outer shell. The Group 7 elements are placed in the vertical column, second from the right-hand side of the periodic table. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
